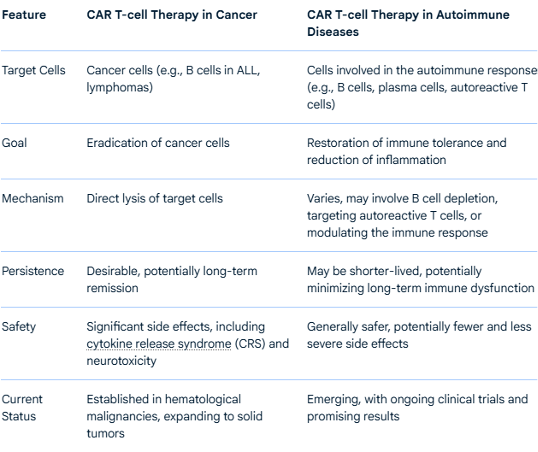
CAR T-cell therapy, a cutting-edge immunotherapy, was initially developed to treat specific blood cancers. However, it is now undergoing clinical trials to explore its potential for treating rheumatic diseases. This procedure involves genetically modifying T cells from a patient to expressing a chimeric antigen receptor (CAR), allowing the T cells to recognize and attach to specific antigens (proteins) on target cells, such as B cells in autoimmune diseases. These CAR T-cells, once activated, can reset the immune system and may lead to remission of the disease. Although CAR T therapy is precise and offers durable outcomes with the possibility of extended remission, challenges remain, including the risk of cytokine release syndrome, immune toxicity, and treatment costs.
CAR T CELL THERAPY FOR CANCER VERSUS AUTOIMMUNE DISEASES
The CAR-T therapy represents a significant advancement in the treatment of autoimmune diseases. Currently, it is being evaluated as a long-term management option due to its effectiveness. This therapy can fundamentally change the immune system responsible for autoimmunity. Unlike cancer treatments, where CAR-T cells remain for an extended period, they are temporarily implanted in patients with autoimmune disease to reduce the likelihood of side effects, infections, and immune dysfunction.
WHY DO WE NEED CAR T THERAPY
In rheumatology, CAR T-cell therapy is important because it offers a fresh treatment approach, especially for challenging cases. CAR T-cells target the immune cells that attack the body, causing inflammation and damage in autoimmune diseases. This could lead to long-term remission. While still under investigation, CAR T-cell therapy has the potential for a lasting reset of the immune system and better outcomes for those with severe rheumatic diseases. For example, CAR T-cells targeting CD19 (a protein found on B cells) in SLE have shown promising results with significant B cell depletion and a potential reset of the immune system.
- Target Autoimmune Conditions:
Rheumatic diseases like systemic lupus erythematosus (SLE), systemic sclerosis (SSc), and antisynthetase syndrome (ASS) arise from issues within the immune system. In these conditions, immune cells attack healthy tissues. CAR T-cell therapy provides a method to specifically target and eliminate these harmful immune cells, potentially slowing the disease.
- Refractory Cases:
Standard treatments like immunosuppressants and biologics often fail for many patients with rheumatic diseases. When traditional methods do not work, CAR T-cell therapy presents a new option. According to the National Institutes of Health (NIH), altering T cells to target specific antigens allows for a more focused and potentially more effective approach to the underlying cause of the disease.
- Possibility of Long-Term Remission:
CAR T-cell treatment for rheumatic disorders aims to restore the immune system over time. This strategy may lead to prolonged disease remission rather than just temporary symptom relief. The treatment seeks to achieve a healthy balance in the immune system by removing autoreactive immune cells. This could significantly enhance patients’ quality of life and lessen the need for ongoing treatment.
- Ongoing Research and Development:
While CAR T-cell therapy is currently used for rheumatic disorders, researchers are working to enhance its effectiveness and safety. This includes exploring different target antigens, improving CAR T-cell design, and refining patient selection criteria. Clinical trials are in progress to assess the long-term impacts of CAR T-cell treatment across various rheumatic diseases.
ADVERSE EFFECTS
Despite its promise for treating rheumatic disorders, CAR T-cell therapy can have side effects. The frequency and severity of these effects can vary based on the specific rheumatic disease being treated and the CAR T-cell construct used.
Common Side Effects:
- Cytokine Release Syndrome (CRS):
During this immune response, CAR T-cells produce a large amount of cytokines, leading to symptoms like fever, fatigue, and, in severe cases, organ damage.
- Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS):
This condition results from immune cells harming the brain, resulting in neurological issues such as seizures, confusion, and symptoms resembling a stroke.
- Long-Term Cytopenias:
CAR T-cell therapy may cause persistently low blood cell counts, particularly in platelets (thrombocytopenia) and neutrophils (neutropenia).
- B Cell Aplasia:
Since CAR T-cells are designed to attack B cells, this can lead to a decrease in the body’s B cell population, affecting antibody production.
- Hypogammaglobulinemia:
Lowered blood antibody levels, or hypogammaglobulinemia, can increase the risk of infections.
- Infections:
Patients may face greater vulnerability to infections because of possible B cell aplasia and immune suppression caused by the treatment.
- Management of side effects:
Monitoring patients and using medications like tocilizumab for CRS and corticosteroids for both CRS and ICANS are standard approaches. Some side effects may be temporary, while others, like B cell aplasia and hypogammaglobulinemia, may persist and require continual monitoring and management.
SPECIFIC RHEUMATIC DISEASES TARGETED BY CAR T-CELL THERAPY
Specifically, CAR T-cell therapy is being explored for systemic lupus erythematosus (SLE), systemic sclerosis, and antisynthetase syndrome. These diseases are marked by excessive B-cell activity and autoantibody production, which CAR T-cells can target.
SLE:
CAR T-cells can penetrate various tissues (such as skin, kidneys, and bone marrow) to deplete B-cells more effectively than some other treatments. Some studies indicate that CAR T-cell therapy can achieve long-term remission in SLE patients, possibly reducing or eliminating the need for other medications. This remission could lessen the dependence on long-term immunosuppressants like glucocorticoids, which can have serious side effects.
Systemic sclerosis:
By targeting B cells and reducing inflammation, CAR T-cell therapy may also help stop or even reverse the fibrotic processes characteristic of SSc. Research is ongoing to evaluate the feasibility and safety of CAR T-cell therapy in SSc patients, particularly those who cannot have hematopoietic stem cell transplantation (HSCT).
Antisynthetase syndrome(ASS):
ASS can be a serious, life-threatening disease, and some patients do not respond to standard treatments like corticosteroids and immunosuppressants, including B-cell-depleting antibodies. Several cases suggest that CAR T-cell therapy can lead to significant improvements in clinical symptoms, such as muscle strength, lung function, and skin lesions, along with reductions in autoantibody levels and the discontinuation of immunosuppressive medications. Some patients have achieved remission without medications.
Other Rheumatic Diseases:
Research is also looking into CAR T-cell therapy for rheumatoid arthritis and vasculitis, though these areas are not as developed.
ENGINEERING CAR T CELLS
CAR T-cell therapy involves genetically modifying a patient’s own T cells to target specific antigens on the surface of autoreactive B cells, leading to their elimination and potentially long-term remission.
- T Cell Collection:
T cells, a type of white blood cell vital for the immune system, are collected from the patient’s blood through apheresis.
- Genetic Modification:
In the lab, these T cells are genetically altered using a viral vector to express a chimeric antigen receptor (CAR).
- CAR Structure:
A CAR is a synthetic receptor that combines an antigen-binding domain (often sourced from an antibody) with intracellular signaling domains from the T-cell receptor.
- CAR Expression and Expansion:
The modified T cells, now expressing the CAR, are then multiplied in the lab to produce a large quantity of CAR T-cells.
- Infusion and Targeting:
The engineered CAR T-cells are infused back into the patient, where they can recognize and bind to cancer cells expressing the target antigen.
- Cell Killing:
Upon binding, the CAR T-cells activate a signaling pathway that leads to their activation, growth, and the destruction of targeted cells.
- Future directions:
Ongoing research aims to enhance CAR T-cell constructs, discover the most effective target antigens, and develop safer, more effective protocols for use in rheumatic diseases.
Authored by:
Dr. Rachel Oommen
MBBS, MD, PDF (Rheumatology)
Consultant Rheumatologist